Publication in Angewandte Chemie International Edition
The Far Side of Carboranes: Anticancer Active Monocations and Ambiently Stable Dications
Authors: Vlastimil Němec, Josef Holub, Maksim A. Samsonov, Zdeňka Růžičková, Josef Cvačka, Ján Vančo, Zdeněk Trávníček, Jan Belza, Zdeněk Dvořák, Jan Vrána, Aleš Růžička
Full-text: https://doi.org/10.1021/acs.inorgchem.5c04198
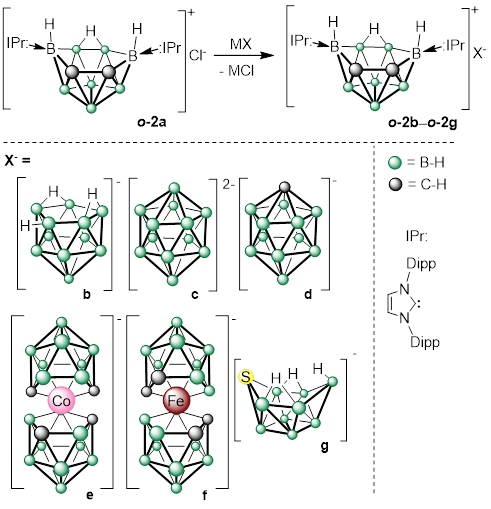
Polyhedral carboranes are highly biologically stable, non-toxic clusters. Whereas they are typically encountered in anionic or neutral forms, positively charged species have only recently been discovered. The in vitro antiproliferative effects of selected carbo- ranes were assessed using a panel of human cancer cell lines, and off-target toxicity was evaluated at normal cell lines. The results demonstrated significant anticancer activity and a favorable resistance factor (RF ≈ 1) for monocationic carborane o-2a, surpassing the effects of doxorubicin and cisplatin. In pursuit of even more efficient substrates, the first dicationic polyhedral boranes were synthesized. These water-stable dications exhibit a reversible closo-/nido- cage opening, triggered either by a strong base (DMAP) or by a combination of triethylamine and molecular hydrogen, and reversed upon the addition of acid. The former transformation proceeds without a redox change, while the latter involves a H2/2H+ conversion in a proton-coupled electron process.